Virtual Twin Guides Life-Saving Heart Surgery
By Sophia Chen
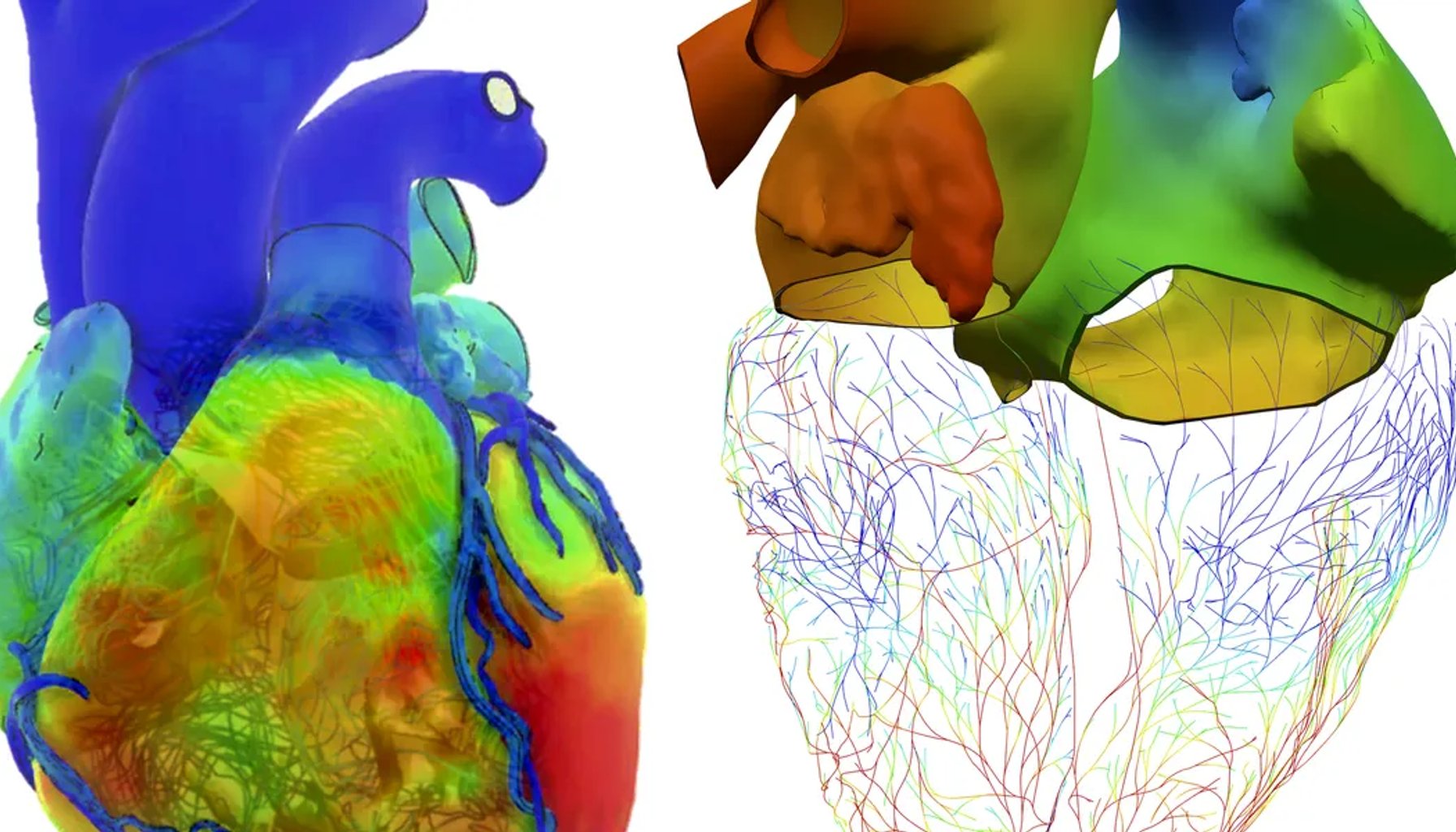
Image / spectrum.ieee.org
A digital twin guided a high-stakes heart operation in a real patient, built from MRIs, CTs, and a careful physics-based model of how this child’s heart actually behaves.
In Boston Children’s Hospital, a cardiac surgeon and a team of engineers stitched together a fully functioning, patient-specific replica of a child’s heart and surrounding vasculature. Over the weeks leading up to the operation, they converted imaging data into a 3D model and then brought the heart “to life” with physics so it could breathe, flow, and flex under stress. The point wasn’t just to see what the heart looks like, but to test strategies long before the first incision. The team could replay dozens of scenario analyses—predicting blood flow, pressure differentials, and muscle-tissue stresses—before deciding the best approach for this exact anatomy.
This is one of the early, real-world demonstrations of a virtual twin in pediatric cardiac surgery—the kind of tool that shifts planning from raw experience to patient-specific physics. Engineering documentation shows the model could mimic the heart’s unique behavior and, crucially, let the surgeon test how a particular cut or stitch would alter outcomes. The larger implication isn’t that doctors will replace judgment with a screen; it’s that the screen can codify patient idiosyncrasies that are easy to miss in traditional planning, where two-dimensional images and the surgeon’s intuition carry most of the weight.
From a practitioner’s lens, several concrete implications emerge. First, the technology leverages actual imaging and a dynamic physics engine to create a “live” simulation of flow and tissue response, not a static blueprint. That distinction matters: fluids in the heart don’t behave like water in a pipe, and myocardial tissue can stiffen or yield differently with each patient. Second, the approach promises a more deterministic preoperative period: if you can try multiple surgical strategies in a synthetic, patient-specific model, you can converge on a plan with fewer surprises in the OR. This represents a meaningful improvement over prior planning that relied primarily on 3D prints or two-dimensional scans. Third, while exciting, the method remains a high-skill, resource-intensive workflow. It demands high-quality imaging, robust computational pipelines, and close collaboration between surgeons and cardio-engineering specialists—elements that don’t scale easily outside well-funded centers.
Yet there are clear boundaries. The experience described in May 2019 reflects a pilot in a single hospital, not a proven, multi-center standard. The predictive power of the twin hinges on imaging fidelity and the physics models’ ability to generalize beyond a lone case. If tissue properties or vascular dynamics shift in unforeseen ways during actual surgery, the model’s forecasts could diverge from reality. In other words, this is a field-ready, real-patient tool in a controlled clinical setting, but not a ubiquitous capability yet. And as with any heavy modeling, there’s a risk of overreliance: clinicians must balance model guidance with intraoperative judgment and real-time data.
Compared with earlier planning methods, this virtual twin marks a substantive leap: it moves planning from static, visual representations toward a dynamic, testable simulation that can quantify how every cut and stitch might reshuffle pressures and flows. It’s not hype—it’s a practical bridge from “what could happen” to “what will happen” for this patient.
Looking ahead, observers should watch for broader clinical validation, cleaner workflows to integrate twins into the OR team, and evidence that multi-patient use reduces corrective interventions or improves outcomes. If the concept scales, hospitals might adopt specialized teams and standardized validation pipelines to turn patient-specific twins into a routine part of high-stakes cardiac surgery.
If successful at scale, digital twins could migrate beyond hearts—into complex vascular surgeries or other organs where flow, tissue mechanics, and individualized anatomy matter as much as the surgeon’s hands. The promise is clear: a data-informed head start that helps surgeons pick the right strategy before the first cut.
Sources
Newsletter
The Robotics Briefing
Weekly intelligence on automation, regulation, and investment trends - crafted for operators, researchers, and policy leaders.
No spam. Unsubscribe anytime. Read our privacy policy for details.